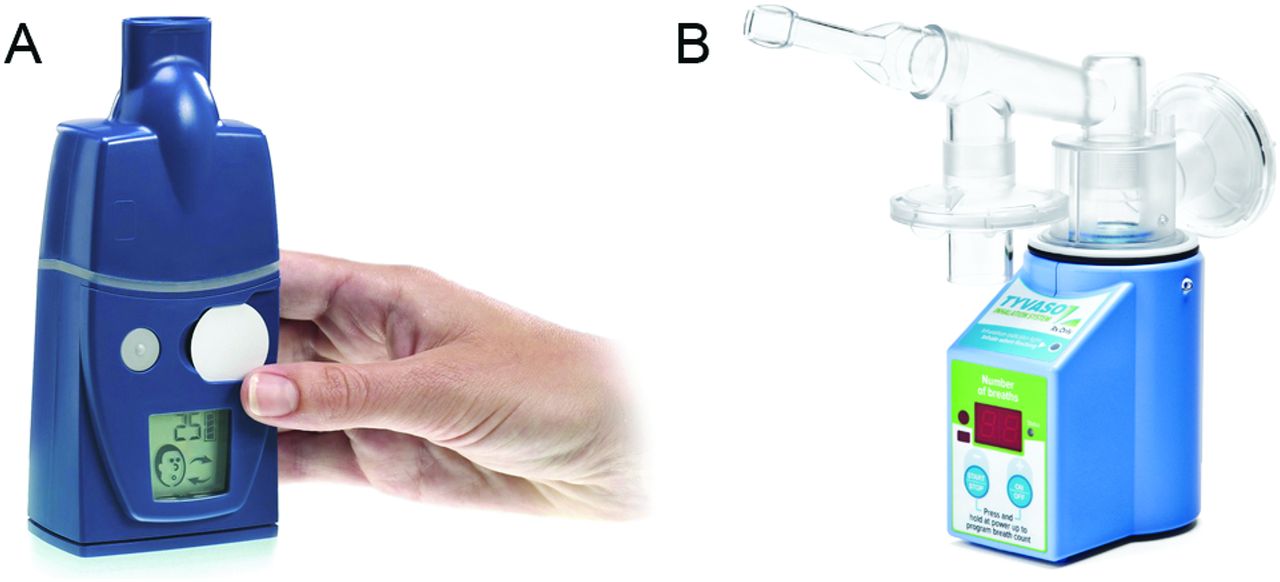
VENTAVIS 10 UG/ML SOLUTION POUR INHALATION PAR NEBULISEUR B/30 AMP DE 2ML | PharmNet - Encyclopédie des médicaments en Algérie | Propriété Sarl ESAHTI

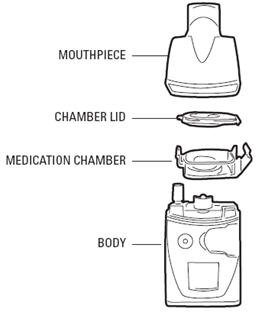
These highlights do not include all the information needed to use VENTAVIS safely and effectively. See full prescribing information for VENTAVIS. VENTAVIS® (iloprost) inhalation solution, for oral inhalation use Initial U.S. Approval: