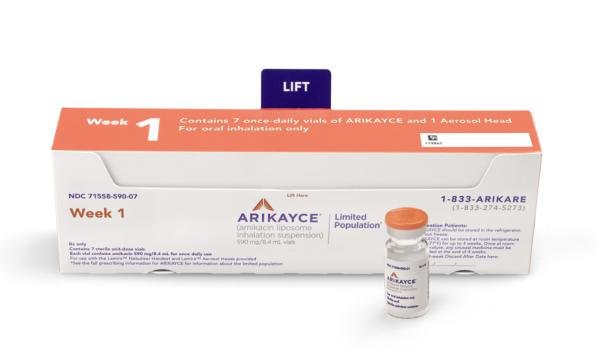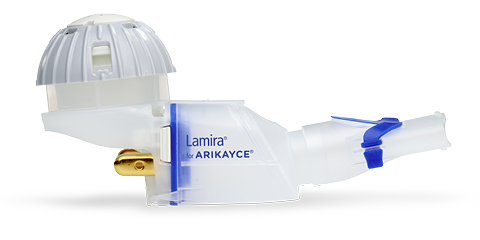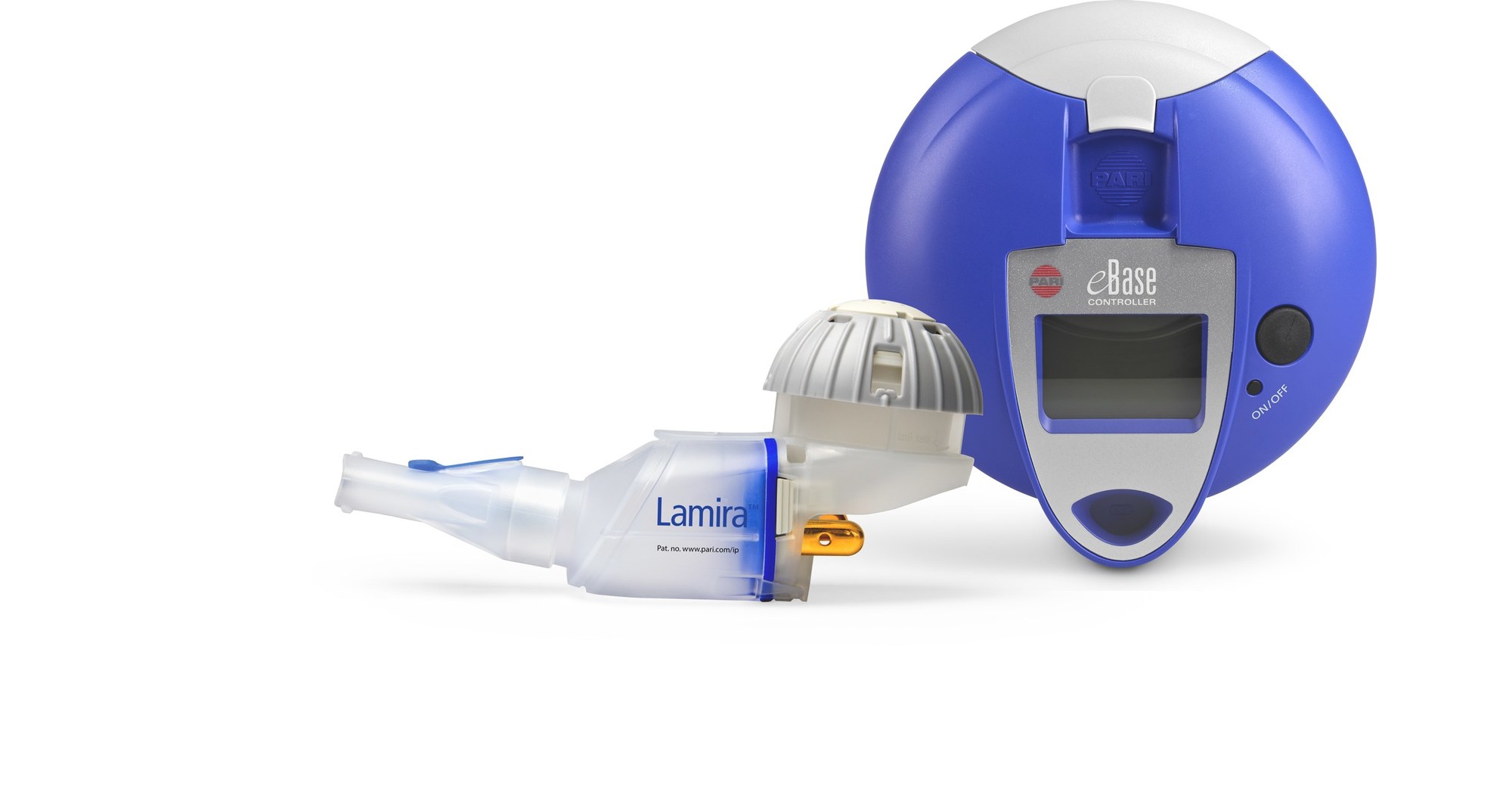
PARI Pharma's eFlow® Technology device, LAMIRA™, approved as the only nebulizer system to deliver Insmed's ARIKAYCE® (amikacin liposome inhalation suspension)

These highlights do not include all the information needed to use ARIKAYCE safely and effectively. See full prescribing information for ARIKAYCE. ARIKAYCE® (amikacin liposome inhalation suspension), for oral inhalation use Initial U.S.
Presentation Title (Insert the Title AAPS approved and that is displayed in the promotional materials for this meeting)